Admission Criteria and Successful Care of Adults in a Tertiary Care Pediatric Hospital During the COVID-19 Pandemic
Objective
The study assessed the health outcomes of adult patients admitted on Pediatric Wards and Intensive Care Unit (PICU) of the Montreal Children’s Hospital (MCH) in Canada during the first and second waves of the COVID-19 pandemic.
Methods
This retrospective chart review analyzed data regarding all adult patients hospitalized at the MCH on pediatric units between March 14, 2020 and March 31, 2021.
Results
Forty (40) adults were admitted to MCH pediatric units. The median age was 28.5 years. There were 26 females and 14 males. The average length of stay (LOS) at the MCH was 6.3 days. There were 32 consultations from adult medical consultants performed at the MCH. Seventeen (17) incident reports were completed. There were no in-hospital deaths, but 6 in-hospital incidents were reported. Within the first seven days post discharge from the pediatric units, 1/24 (4%) patients returned to the adult emergency room and an additional 4 patients (16,6%) returned within 30 days.
Conclusion
With clear admission criteria, careful planning, and well-planned supportive resources treating adult patients within a pediatric care facility by pediatric care teams, also caring for pediatric patients in the same units, could be considered as a safe contingency plan in a time of crisis. Furthermore, admitting and treating adult patients on several pediatric units (PICU) may provide a larger variety of options for admission of adult patients.
Introduction
The COVID-19 pandemic put a significant stress on the healthcare systems leading to a shortage of hospital beds and equipment, disproportionally affecting older adults compared to the pediatric population1. In response to the critical rise in adult care demands, and considering that very few children required hospitalization due to COVID-19, innovative collaborations were developed to expand adult care capacity.
Pediatric medical staff from children’s hospitals and pediatric centres worldwide stepped up to support their adult colleagues. Pediatric institutions employed different strategies to address this challenge, including the admission of adults on pediatric care units 2,3. Others adopted a hybrid model of care 4-6, or entirely repurposed their units to exclusively care for adults 7-9. In most cases, pediatric patients were either consolidated onto other units within the same hospital, transferred to standalone pediatric hospitals, or referred to community centres when specialized care was not required.
The first COVID-19 positive case in Canada was reported on January 25, 2020, and on February 27, 2020 in Quebec10-11. On March 14 of the same year, the Quebec government declared state of health emergency12. Rapidly, Montreal became the epicentre of COVID-19 in Canada with the most cases during the first wave13. The Montreal Children’s Hospital (MCH), part of the Glen site of the McGill University Health Center (MUHC) was the first Canadian pediatric centre to admit adult patients within its own walls during the COVID-19 pandemic.
There is limited knowledge on the occurrence of adverse events for adult patients admitted in pediatric hospitals, and on adult patients being treated by hospitalist pediatricians in Canada.
The objective of this study was to assess outcomes of adult patients admitted on pediatric wards and intensive care unit (PICU) of the Montreal Children’s Hospital (MCH) during the first and second waves of the COVID-19 pandemic.
Methods
Study Design and Setting
In this retrospective study, a thorough medical chart review of adult patients hospitalized on the MCH units (PICU, pediatric medicine, and pediatric surgery) during the first and second wave of the COVID-19 pandemic between March 14, 2020 and March 31, 2021 was conducted. The first wave occurred between March 14, 2020 and August 31, 2021 whereas the second wave took place from September 1, 2021 through March 31, 2021. The MCH is an urban tertiary care pediatric hospital and one of the four physically joint care facilities that form the MUHC, which is situated in Montreal, Canada. Admission criteria were developed carefully during the pandemic by a multidisciplinary group of pediatric and adult health providers and were used to select the adults to be admitted on pediatric units (Table 1).
Data Collection
Medical charts were reviewed by one researcher. The data was recorded in an excel spreadsheet, specifically designed for this study. The collected data included: patient demographics, primary diagnosis (as reported on the hospitalization summary), length of stay, location of the hospitalization, number of consultants from the adult medical staff involved, whether transfers occurred at any point during hospitalization on pediatric units (from pediatric wards to PICU and from pediatric units to adult units), disposition, and patient health outcomes (including: Code Blue activations, in-hospital deaths, incident reports, unplanned return visits to any of the MUHC emergency departments).
Data analysis
The study is descriptive, and no comparison groups were included. Descriptive statistics, encompassing the mean, standard deviation, and interquartile range (IQR), were computed to analyze various aspects of the dataset, such as patient age, length of stay and the involvement of adult consultants. Additionally, frequency tabulations were utilized to analyze primary diagnoses and patient health outcomes such as transfers, code blue activations, in-hospital deaths, incident reports, dispositions, and unplanned return visits.
Ethical Considerations
The MUHC Research Ethics Board approved the study protocol (#2022-7761) which was conducted in accord with the Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans 2 (2018), as well as in respect of the requirements set out in the applicable standard operation procedures of the MUHC Research Institute. Anonymity and confidentiality were maintained throughout the study.
Results
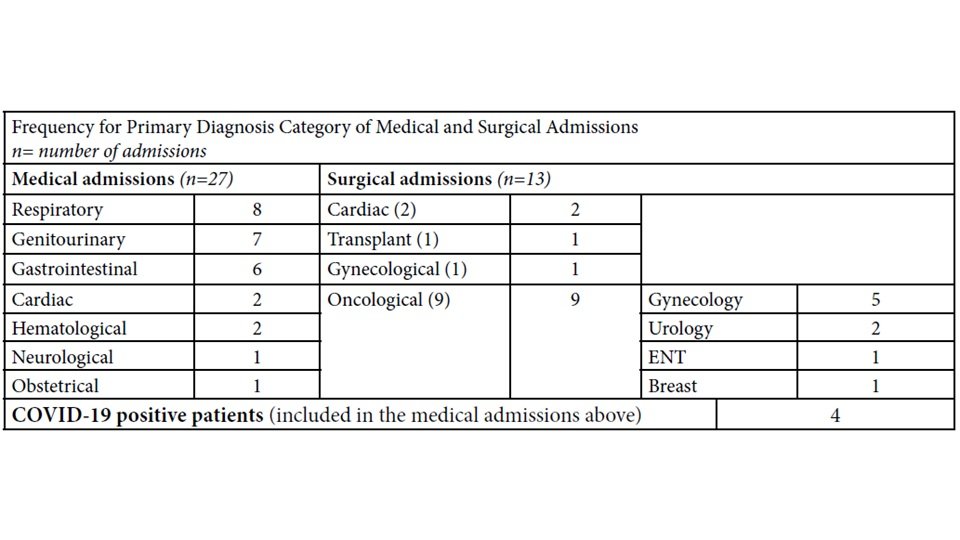
This retrospective chart review identified a total of 40 admissions of adult patients to the MCH pediatric medical and surgical wards, as well as the PICU. These patients were cared for by a pediatric team on shared units alongside regular pediatric patients. The patients were mostly young, healthy adults, with a median age of 28.5 years. Among them, 26 were females and 14 were males ( Table 2). The average length of stay for all adult patient admissions was 6.3 days at the MCH and 22.2 days overall for those who were transferred back to the adult site when bedspace became available to complete their hospitalization.
As demonstrated in Table 2, the 40 adult patients included in the study received a total of 132 consultations from adult medical consultants, with the number of consultations per patient ranging from 0 to 16. Of note, only 32 consultations occurred during the hospitalization on the pediatric units, divided amongst 22 patients. Among those, 15 consultations were in follow-up to the initial non-MCH phase of admission, while 17 were initiated by the pediatric medical staff to address evolving healthcare needs of patients during their stay on the pediatric units.
Seventeen (17) incident reports were completed during admission at the MCH. There were no in-hospital deaths reported, and 6 in-hospital complications occurred: 1 line infection; 1 code blue activation; 1 accidental extubation, and 3 cases of ventilator-associated pneumonia.
Following discharge from the MCH, most adult patients (n=24) returned home, while others were transferred to various healthcare facilities (n=16). Within 7 days post discharge from the pediatric units, 1 out of 24 patients (4%) returned to the adult emergency room, and an additional 4 patients (16.6%) returned within 30 days. Reasons for return visits varied and included new or worsening symptoms, post-operative complications, therapeutic interventions, or equipment dysfunctions. These findings are detailed in Table 4.
Discussion
A MUHC-wide strategy was implemented to provide the necessary capacity for adult patients while continuing to care for pediatric patients. During the first and second waves of the pandemic, this strategy involved admissions of adult patients on the PICU as well as the pediatric medicine and surgical wards of the MCH.
The MCH opted to implement a hybrid model of care, wherein adults received treatment alongside children, administered by the same pediatric medical staff and within the same facility. Admissions originated from various sources and encompassed diverse primary diagnoses, resulting in positive and safe patient health outcomes. A pivotal factor contributing to this success was the collaborative development of clear admission criteria. Indeed, these criteria were developed by a multidisciplinary committee mandated to elaborate patient care trajectories and admission guidelines for adults on the different pediatric units ( Table 1). The selection of adult cases that were not excessively complex ensured favorable health outcomes for these adult patients admitted to pediatric units while also ensuring safety of the admitted pediatric patients.
The study by Deep & colleagues4 described solely a hybrid pediatric and adult critical care unit without involvement of other pediatric units. The identification of adult patients for admission to the hybrid critical care unit was determined by a tactical lead from the adult team who would discuss individual patients for admission with the pediatric critical care lead. No predetermined admission criteria were reported.
Sinha et al reported that 7 PICUs in England repurposed their space, equipment, and staff to care for a total of 145 critically ill adults during the first wave of the pandemic. The adults admitted were older (median age ranging from 57 to 62 years old in the different PICUs) as well as their in-patient mortality rate. Indeed, 20 of 145 (14%) adult patients admitted died in the PICU vs 0 in-patient deaths in all the pediatric units caring for adult patients at the Montreal Children’s hospital. In addition, the study did not publish any specific admission criteria for adults care for the 7 PICUs.
Our experience at the MUHC reported in this study highlights both the effectiveness and safety of pediatric teams caring for adult patients in the PICU, as well as on pediatric surgical and medical wards. Collaboration and communication between the pediatric and adult medical teams as well as careful elaboration of admission criteria were instrumental in the success of the strategy.
Whilst being very safe, the strict criteria established that adult patients infected with COVID-19 and requiring ICU level care had to be aged 30 years old or less to be considered for admission on the PICU. This significantly limited the number of potential adult PICU admissions. Return visits to the emergency room after discharge were only assessed at the MUHC and not in other nearby hospitals. In addition, the findings of this study may not apply to adult and pediatric facilities that are not located inside the same building. In such contexts, support from adult health providers may not be readily available and transport of patients from one hospital to the other may cause delays to admission.
Conclusion
With clear admission criteria, careful planning, and well-planned supportive resources, treating adult patients within a pediatric care facility by pediatric care teams, also caring for pediatric patients in the same units, could be considered as a safe contingency plan in a time of crisis. Furthermore, admitting and treating adult patients on several pediatric units (PICU, pediatric medicine ward, and pediatric surgical ward) may provide a larger variety of options for admission of adult patients. It would be ideal for healthcare facilities, without such experience, to prepare a similar strategy before the next pandemic strikes. Future research could assess if a similar strategy would be safe in other contexts, such as mass casualty incidents, natural, or manmade disasters.
Acknowledgements:
Additional contribution: We extend our appreciation to David Iannuzzi for providing research assistance during data collection, Karine Jones for her support in manuscript preparation, and Lindsay Hales for her excellent librarian support.
Declarations
Author contributions
All authors contributed equally and validated the final version of record.
Conflicts Of Interest
The Author(s) declare(s) that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Registration
No registration applicable
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical approval
Ethical approval for this study was not required.
References
1. Guan W, Ni Z, Hu Y, Liang W, Ou C, He J, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020 Apr 30;382(18):1708–20. https://doi.org/10.1056/NEJMoa2002032
2. Dong Y, Mo X, Hu Y, Qi X, Jiang F, Jiang Z, et al. Epidemiology of COVID-19 among children in China. Pediatrics. 2020 Jun;145(6):e20200702. https://doi.org/10.1542/peds.2020-0702
3. Lu X, Zhang L, Du H, Zhang J, Li YY, Qu J, et al. SARS-CoV-2 Infection in Children. N Engl J Med. 2020 Apr 23;382(17):1663–5. https://doi.org/10.1056/NEJMc2005073
4. Deep A, Knight P, Kernie SG, D’Silva P, Sobin B, Best T, et al. A Hybrid Model of Pediatric and Adult Critical Care During the Coronavirus Disease 2019 Surge: The Experience of Two Tertiary Hospitals in London and New York. Pediatr Crit Care Med. 2021 Feb;22(2):e125–34. https://doi.org/10.1097/PCC.0000000000002584
5. Sinha R, Aramburo A, Deep A, Bould EJ, Buckley HL, Draper ES, et al. Caring for critically ill adults in paediatric intensive care units in England during the COVID-19 pandemic: planning, implementation and lessons for the future. Arch Dis Child. 2021 Jun 1;106(6):548–557. https://doi.org/10.1136/archdischild-2020-320962
6. Osborn R, Doolittle B, Loyal J. When Pediatric Hospitalists Took Care of Adults During the COVID-19 Pandemic. Hosp Pediatr. 2021 Jan 1;11(1):e15–8. https://doi.org/10.1542/hpeds.2020-001040
7. Yager PH, Whalen KA, Cummings BM. Repurposing a Pediatric ICU for Adults. N Engl J Med. 2020;382(22):e80. https://doi.org/10.1056/NEJMc2014819
8. Philips K, Uong A, Buckenmyer T, Cabana MD, Hsu D, Katyal C, et al. Rapid Implementation of an Adult Coronavirus Disease 2019 Unit in a Children’s Hospital. J Pediatr. 2020 Jul 1;222:22–7. https://doi.org/10.1016/j.jpeds.2020.04.060
9. Joyce CL, Howell JD, Toal M, Wasserman E, Finkelstein RA, Traube C, et al. Critical Care for Coronavirus Disease 2019: Perspectives From the PICU to the Medical ICU. Crit Care Med. 2020;48(11):1553–5. https://doi.org/10.1097/CCM.0000000000004543
10. Berry I, Soucy JPR, Tuite A, Fisman D. Open access epidemiologic data and an interactive dashboard to monitor the COVID-19 outbreak in Canada. CMAJ. 2020 Apr 14;192(15):E420. https://doi.org/10.1503/cmaj.75262
11. Santé et Services sociaux. Cas confirmé de COVID-19 au Québec [Internet]. Quebec: Gouvernement du Québec; 2020 [cited 2021 Mar 1]. Available from: https://www.quebec.ca/nouvelles/actualites/details/cas-confirme-de-covid-19-au-quebec
12. Santé et Services sociaux. Pandémie de COVID-19 - Le gouvernement du Québec déclare l’état d’urgence sanitaire, interdit les visites dans les centres hospitaliers et les CHSLD et prend des mesures spéciales pour offrir des services de santé à distance [Internet]. Quebec: Gouvernement du Québec; 2020 [cited 2021 Mar 1]. Available from: https://www.quebec.ca/nouvelles/actualites/details/pandemie-de-covid-19-le-gouvernement-du-quebec-declare-letat-durgence-sanitaire-interdit-les-visites-dans-les-centres-hospitaliers-et-les-chsld-et-prend-des-mesures-speciales-pour-offrir-des-services-de-sante-a-distance
13. Health Canada [Internet]. Quebec: Government of Canada; 2020. Epidemiological summary of COVID-19 cases in Canada. Available from: https://health-infobase.canada.ca/covid-19/epidemiological-summary-covid-19-cases.html