The Assessment of Lebanese Hospital Nurses’ Qualifications in The Fight Against COVID-19: Are They Well Prepared?
Background
On December 31, 2019 China reported multiple cases of a novel coronavirus, This rapid and unexpected increase in cases caused a shortage of testing kits, personal protective equipment (PPE), medical treatments, ventilators, and other medical supplies. Therefore, nurses and physicians needed to feel safe, protected and confident while treating ill patients. It is the responsibility of the healthcare system's to wisely use the skills and available resources for an effective response and ensure the safety of the personnel. Our primary objective is to assess the preparedness of nurses against the coronavirus and then to evaluate the impact of the outbreak on the nurses and their attitude towards the pandemic.
Methods
An anonymous online survey was conducted among nurses across Lebanon. It consisted of 24 multiple-choice, closed-ended questions related to knowledge, attitude and practice.
Results
A total of 370 registered nurses completed the survey. Analysis showed that age had no effect on knowledge; males were more trained than females for triage, hand hygiene and working in isolation units, while females were more trained for handling PPE. Additionally, females attended national lectures more than males, while males received information from media and brochures. The training for PPE use and working in isolation unit was better conducted in university hospitals as compared to non-university ones. 50% of nurses in both hospital types did not attend any lectures. The nurses in all Lebanese districts had training about hand hygiene and working in isolation unit before the pandemic while they had the triage training mostly after the pandemic started. Analysis also showed that the nurse-to-patient ratio was higher for older nurses and in peripheral areas. For both genders, 66.3% had never undergone an N95 fit test. Lastly, almost half of the nurses thought about quitting their job.
Conclusion
The study showed that there is some lack of nurses’ knowledge towards COVID-19. Variations were observed in nurses’ practice regarding age, gender, hospital type, district and nurses’ position.
Introduction
On December 31, 2019 China reported multiple cases of acute respiratory illnesses that were later found to be caused by a novel coronavirus, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. This virus is a member of the subfamily Coronavirinae and belongs to the same genus as coronaviruses that caused the severe acute respiratory syndrome (SARSCoV) reported in China in 2003, and Middle East respiratory syndrome (MERS-CoV) reported in Saudi Arabia in 2012 [2]. Unfortunately, the new coronavirus had spread very rapidly around the globe, so on March 11, 2020 the World Health Organization (WHO) declared SARS-CoV-2 a pandemic [3]. This rapid and unexpected increase in cases caused a shortage of testing kits, personal protective equipment (PPE), medical treatments, ventilators, and other medical supplies.
In addition, the insufficiency was not only observed in material resources but in human resources as well, number of staff and preparedness of personnel [4]. Despite the elapse of more than a year after the declaration of the pandemic, there is still no curative treatment for SARS-CoV-2 and numbers continue to rise with nearly one million new cases in the last week of March 2021 and the emergence of new strains, more virulent, like the latest one in India [5-6]. According to Guest et al, the major steps in overcoming this pandemic are an effective lockdown, an increase in testing and most importantly an improvement of the healthcare system’s capacity to respond, which is the aim of this study [7]. In turn, ensuring the safety of health care workers (HCWs) should be a priority by vaccinating them, providing them with the appropriate PPEs, giving them the adequate training and updating their knowledge of the pandemic.
Researchers in Taiwan found that the number of HCW decreased as the sense of security among them decreased; they felt unsafe treating coronavirus disease (COVID-19) patients, and avoided working in COVID-19 units [3]. Therefore, nurses and physicians needed to feel safe, protected and confident while treating ill patients; for this purpose, infectious disease specialists in Taiwan developed a “Traffic control bundling” map that ensures a passage for patients from triage to treatment zone where HCW are not exposed to the patient and those who are directly taking care of them are in full PPE. This map aims at preventing shortage in medical staff secondary to anxiety, thereby improving the workflow [3]. It is also very important for the workflow that the medical staff in a pandemic is not subject to any anxiety or stress. while it is difficult to determine the number of lives saved by epidemiological research, yet it is certain that good intervention and prevention strategies save millions of lives.
It is our responsibility to wisely use the skills and available resources for an effective response. For this reason, the WHO published a checklist to help hospitals assess their preparedness in order to ensure a rapid response to the outbreak [8]. This study aims to assess how well the Lebanese nurses are prepared for this new pandemic. Our primary objective is to assess the preparedness of nurses against the SARS-CoV-2 outbreak based on the level of knowledge, training and practice. The secondary objective is to evaluate the impact of the outbreak on the nurses and their attitude towards the pandemic.
Materials and methods
Study design and data collection procedure
This is a descriptive, observational cross-sectional study, for which the approval of the Institutional Review Boards of the Lebanese American University was obtained in May 2020 IRB #: LAU.SOM.RS2.11/ May/2020. It was based on an anonymous self-administered online survey. The questionnaire was created using Google Forms, saved as a link, and reviewed by the Lebanese Order of Nurses who distributed it via email to all registered nurses. Data collection took place between July 2020 and February 2021 (The Beirut Blast that occurred on the 4th of August 2020, caused a halt in data collection for approximately two months.) Only answers of currently practicing nurses, whether in the private or public sector, working in any of the Lebanese governorates, were included. Nurses who were not registered, did not hold a license to practice, or were not currently practicing were excluded from the study.
Sample size
The sample size was calculated using Epi Info 7 StatCalc functions for a population survey. Given that the number of registered nurses in 2020 was 16927 and, with an expected confidence level of 95% along with a margin of error of 5%, the minimum sample size required to obtain statistically significant results was determined to be 370 nurses.
Survey development
The questionnaire was designed after reviewing multiple studies discussing COVID-19 preparedness among HCW, as well as preparedness for other epidemics and pandemics. It was initially written in English then translated to Arabic by a translator and cross-checked with the team of the study. It was then back-translated to ensure accuracy to English. Both the English and Arabic versions were piloted by two nurses, one day apart, to make sure they were equally understood. This way we were able to check for the comprehension of the questionnaire in both languages and agreement (reliability of the questionnaire) in their answers before conducting the study. The questionnaire was composed of 24 multiple choice questions that mainly look into the sociodemographic characteristics of the nurses, their training and educational backgrounds. This allowed us to assess nurses’ knowledge, the load they were dealing with and to analyze their practice. In addition, there were few questions that look into nurses’ attitude towards this crisis. To note that the survey was sent in both languages, Arabic and English, and the answers provided were collected on an online sheet.
Data management and statistical analysis
Statistical analysis was conducted using the Statistical Package for the Social Sciences (SPSS) for Windows, Version 23.0. Means and standard deviations were reported for continuous variables. Categorical variables were assessed and described as frequency and percentage. Bivariate analysis was performed using Chi square test and Fisher test to examine associations between knowledge, practice and attitude and the different sociodemographic variables (gender, age, experience, job position, years of experience, type of hospital and hospital location). A p-value of <0.05 was considered statistically significant.
Results
General Characteristics
A total of 370 nurses responded to the questionnaire; however, only 359 were currently practicing and thus included in the study. Among them, 155 were male (41.8%) and 215 were female (58.2%).Respondents’ ages ranged from 21 to 63 with a mean age of 32 years. Additionally, 223 (62.1%) worked in private hospitals, 89 (24.8%) worked in public hospitals and 47 (13.1%) worked in both sectors simultaneously. When considering the type of hospital, 204 (56.8 %) participants were practicing in university hospitals while the rest were working in non-university hospitals. Moreover, when considering the geographical distribution, the majority 167 (46.5 %) of nurses were practicing in the capital Beirut, and the rest were distributed across the North, South, Bekaa and Mount Lebanon.
After reviewing the numbers and the descriptions of the sample obtained, we tried to observe the correlation between the above-mentioned demographics and other characteristics such as experience, job position…with the knowledge, practice and attitude of the nurses towards the pandemic; result of our observations is described in the following section.
Nurses’ Knowledge
To assess nurses’ knowledge, the questionnaire included questions regarding training (triage of COVID-19 patients, use of PPEs, hand hygiene and different types of isolation) and educational activities (such as conferences’ and lectures’ attendance), were included in the questionnaire. Regarding training, we looked into possible associations with participants’ age and gender, also with the type and location of hospital.
Association with age
Data analysis showed no association between age and any of the previously cited variables. Therefore, age had no effect on nurses’ knowledge of SARS-CoV-2.
Association with gender
Data analysis showed an association between gender and studied variables, as summarized in Table 1.
Male nurses had more training sessions than females, in triage, hand hygiene and working in isolation units. In contrast, female nurses received more training in the proper use of PPE. Our observations found that nurses were trained for triage and PPE handling after the emergence of the SARS-COV-2 in Lebanon, while trainings concerning hand hygiene and different isolation types, were acquired before the start of the pandemic. To note that a small percentage of nurses never had any type of training (around 16 % for triage training and 8% for each of the rest).
Concerning the information platforms, 27% of respondents reported access to international resources such as those from UNICEF or WHO and 79% noted receiving information from infection control committee of their hospital. However, no statistically significant difference was observed between genders for the abovementioned sources. 30.9% of the nurses received information from the national health authorities (Ministry of Public Health), with a predominance among female respondents. 70.8% of nurses received their information from media, 49% from brochures and 42.3% from occupational health and safety committees; with a significant male predominance across all three sources.
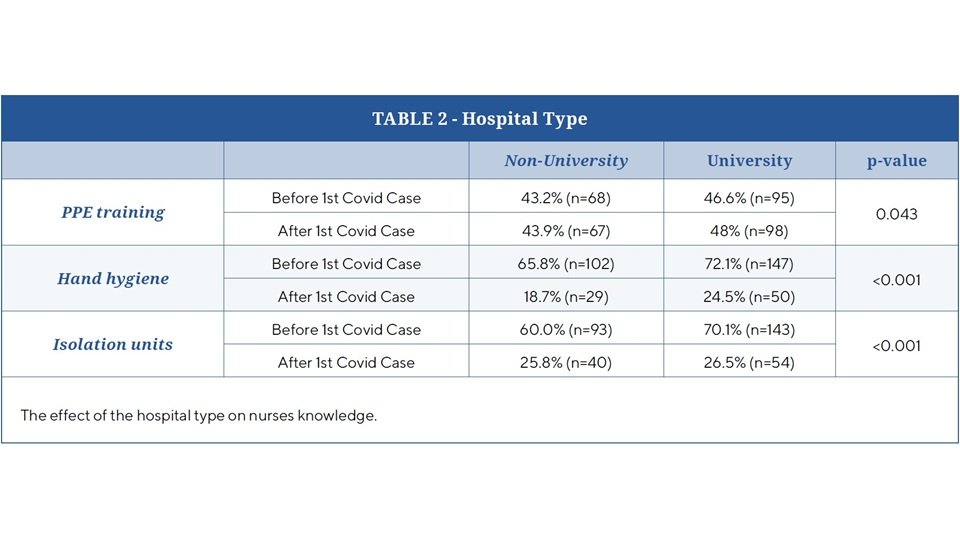
Association with type of hospital (University vs. Non-University hospital)
The data is summarized in Table 2.
For triage training, data analysis showed no significant difference between university and non-university hospitals. As for the rest, before and after the pandemic there was a statistical difference concerning training for PPE use (p value 0.042), hand hygiene (p < 0.001) and working in isolation units (p< 0.001) was significantly more common in university hospitals.
It is important to note that not all hospitals provided training sessions. In fact, 8% of hospitals in both categories offered no training at all.
As for taking lectures, data was significant for the following observation: 35% of nurses working in university hospitals attended lectures from International Organizations as compared to 16% of nurses working in non -university hospitals. 37 % of nurses working in university hospital attended lectures given by the Ministry of Public Health while only 23% of those working in non-university hospitals did. Almost 50% of nurses from both hospital types did not attend any lectures. On the other hand, 62 % of nurses working in non-university hospitals relied more on self-education through random brochures compared to 32% of those in university hospitals.
Association with hospital location in Lebanese governorates
Training for hand hygiene and working in isolation units, were already implemented in hospitals across all Lebanese governorates. For triage, triage training was introduced after the pandemic began.
As for the PPE, For PPE training, it was observed that hospitals in Beirut and Mount Lebanon enforced training mainly after the emergence of the first confirmed case in Lebanon.
Regarding lectures, nurses working in Beirut and Mount Lebanon area attended more international and national lectures, as compared to nurses working in Bekaa, North and South districts. Nurses in the latter regions were mainly getting their information from media and brochures. In all the districts, the hospital infection control and the occupational health and safety committees played an equally active role in educating nurses.
Association with nurses’ position and years of experience
Nurses' roles were categorized into three groups, head nurses, critical care nurses (Emergency Room and Intensive Care Unit) and non-critical nurses (Pediatrics and regular ward). All lectures and trainings were not significantly affected by position. However, 22% of non-critical care nurses, 5% of head nurses and 14% of critical care unit nurses never had triage training. Neither getting any training nor attending lectures showed a statistically significant difference when comparing years of experience.
Nurses’ Practice
Using the same variables (gender, age, hospital type, hospital location, and job position), the practice of nurses was evaluated. The key indicators assessed were whether nurses underwent N95 fit testing, their experience working in negative pressure units, and the patient load assigned to them.
Association with age
Data analysis showed that no significant difference was observed in N95 fit testing when comparing different age groups, nor in working in negative pressure units. However, the assigned number of patients increased as nurses’ age increased. Actually, nurses who were 30 years and older had 10 to 20 patients to take care of whereas nurses who were 30 years and younger had 5 to 10 patients to take care of.
Association with gender
For both genders, the overall rate for N95 fit testing was very low: in total 66.3% of nurses reported never having undergone an N95 fit test. Within those who had the test, 30.1% of the females and 18% of males had the fit test after the pandemic while 8.1% of females and 9% of males had it before. Regarding working in negative pressure unit, male nurses significantly had previous experience, 39.3% of males as compared to 28.7% of females (p=0.035). As for the load of patients during their practice there was no significant difference between genders.
Association with type of hospital (University vs non-university)
Hospital type (university vs. non-university) did not seem to affect the practice of the nurses. Association with hospital location in Lebanese governorates Beirut was the area where nurses had N95 fit test done the most, and mainly after the start of the pandemic (31.7%, or n=53 with p-value =0.002). The load of the patients assigned to each nurse during the pandemic was higher in peripheral areas (North, South and Bekaa) as compared to the Capital, however, no statistical difference existed before the pandemic between the different districts.
Association with nurses’ position
66% (n=238) of the of nurses did not have the N95 fit test done at all, 8% (n=31) did it before the pandemic
and 26% (n=90) after. 42% (n=51) of critical care nurses were familiar with working in negative pressure unit compared to non-critical care and head nurses (28.6 % or n=68).
Nurses’ Attitude
To understand the emotional and psychological burden experienced by Lebanese nurses during the COVID-19 pandemic, the last section of the survey contained two questions that address the nurses’ will to take an unpaid leave or even quit their job. A bivariate analysis of the different studied characteristics (age, gender, hospital type and location, and job position) showed that none of them was significantly associated with the nurses’ will to take a break or quit. In all categories, almost 50% of the respondent nurses thought of taking an unpaid leave, the percentage decreases to 33% of the total responses when asked about quitting their jobs.
Discussion
HCW and in particular nurses are at the frontline of SARS-CoV-2 pandemic response. In this study we assess the knowledge, practice and attitude of the Lebanese nurses towards the pandemic all over Lebanon.
Knowledge is a key factor in the fight against any pandemic, since a poor understanding of the disease can result in delayed identification of positive cases and therefore lead to rapid spread of the infection.
Guidelines for HCW along with online courses have been developed by several national and international organizations such as the Lebanese Ministry of Public Health, infectious disease societies, the WHO, the Centers for Disease Control and Prevention, and the National Institutes of Health [9].
This is not the first study to explore the awareness and attitude of Lebanese HCW in Lebanon regarding the pandemic. Other studies done earlier, only described the knowledge and attitude of nurses and pharmacists in Lebanon but without trying to identify the reasons behind the deficiency and consequently failed to provide a space for improvement [10].
In a similar study, Saade et al. analyzed the knowledge and attitude of Lebanese nurses and reported that they were well informed, based on their understanding of the disease transmission, clinical presentation and effective protective measures [10].
However, our study focused more specifically on the source of this knowledge and we found that the majority of nurses (almost 2/3) did not receive any lectures or trainings from international organizations while the study of Saade et al showed that ¾ (74.6%) attended an awareness session on COVID-19. The information gathered by the nurses was from personal effort in looking at various accredited and non-accredited resources online, from media and brochures, especially in non-university hospitals and outside the capital [10]. This means that a big number of Lebanese nurses are getting their information from non-evidence-based sources, which raises concerns about the precision of this knowledge. A study conducted in Lebanon by Domiati et al. found that 61% of the Lebanese population obtained their COVID-19 knowledge from HCWs, positioning nurses as a key source of public information [11]. The Lebanese Order of Nurses could play an important role in solving this issue, by helping the registered nurses in attending virtual international and national conferences, based on evidence- based, updated guidelines and on experts’ opinion. Hospitals also have a responsibility to promote the role of infection control and occupational health and safety committees in educating their nurses and improving their trainings. Although the Lebanese authorities including the Ministry of Public Health launched a dedicated website for all information regarding COVID-19 and started broadcasting preventive methods on local television channels. These efforts primarily target the general population, and not HCWs [12].
The Ministry of Public Health has an important part to play through updating daily news, about the cases in Lebanon, and awareness campaigns. Finally, the nurses themselves should also be encouraged to read articles from trusted and evidence-based literature.
In most similar studies, males demonstrated low levels of knowledge, attitude and preventive practices compared to females [13-16]. However, in our study male nurses received more training and lectures. This may be due to some hospitals assigning male nurses to SARS-CoV-2 wards more frequently and investing more in their training. It could also be secondary to some societal norms favoring educational opportunities for men over women.
Similar to finding of a study looking at Lebanese pharmacists’ practice towards the pandemic [17]. Our results showed that nurse’s practice was also affected by not having enough PPE, and also by not having N95 Fit test, which can render the nurses hesitant in their jobs. The delivery of PPE is a must to provide adequate protection of healthcare workers and other non-infected patients.
Compared to Saade et al, our questionaries’ regarding practice was complementary to theirs [10]. They asked about getting the infection, being tested for COVID 19, having infected co-workers, treating COVID 19 patients, having sufficient PPEs and correctly using it, evaluating hospital preparedness, educating patients and testing the n95 masks themselves (46% vs 34% in our study) [10]. Our study addressed, on top of the fit test, the load of patients per nurse and working in negative pressure units. The load per nurses was observed in older nurses, probably because they are more experienced and have greater resilience. Moreover, the nurse-to-patient ratio was greater for those working in peripheral areas most probably because of the lack of staff working in rural areas, and the lower number of hospitals as compared to the capital. This finding is comparable to a similar study done in China where the nurses in urban areas had more knowledge especially the ones with more than 20 years of experience and therefore were assigned a higher patient load [18].
Finally, when assessing the attitude of nurses, Saade et al. described Lebanese nurses as being afraid of getting the infection, or transmitting it to their loved ones [10]. Although a study done in the Philippines showed that the fear of getting infected with COVID-19 is associated with higher knowledge and that is the case with nurses since they have more knowledge of the disease than the general population [19]. Saade et al. also documented the nurses’ psychological burden by looking at their levels of anxiety, stress and depression, and the negative consequences of these burdens on their duties, relationship with their families and co-workers [10]. Saade et al. focused on the psychological impact on the nurses’ attitude; one common question between the two studies, regarding leaving work due to COVID, showed similar results: 64% of their respondents’ vs 67% of ours declared not wanting to quit. This actually shows a positive attitude toward COVID-19 and the result is comparable to the study done in the general population in Lebanon [11]. This reasoning can also be explained by the economic crisis affecting the country and further studies about nurses’ burnout should be addressed.
Limitations
The study was conducted through an online survey therefore only nurses who had access to it were able to complete it. Also, since the data are based on self-reported responses, there might be information and recall bias. Since the study is cross-sectional, we cannot conclude any direct causality between dependent and independent variables. More specifically, our survey only addressed some aspects of knowledge, practice and attitudes; we therefore didn’t assess the reasons behind nurses’ will to quit or take unpaid leaves.
Conclusions
This study showed gaps in knowledge regarding SARS-CoV-2 among Lebanese nurses, mainly due to the absence of a proper educational program and training. This gap persisted despite the fact that the COVID-19 pandemic had been ongoing for over 6 months during the study period. However, variations existed in nurses’ practice regarding age, gender, hospital type, district and nurses’ position. The nurses, despite the pandemic, did not report a high willingness to quit or take unpaid leaves.
In conclusion, greater efforts must be invested in educating and training nurses, who represent the backbone of the healthcare system. These efforts should be coordinated by multiple stakeholders, including scientific societies, the Lebanese Order of Nurses, and the Ministry of Public Health. Moreover, hospitals should be adequately equipped with essential resources to face a pandemic. This moment offers a valuable opportunity to reassess outbreak management challenges and develop sustainable strategies for future health crises. Additional studies are also recommended to evaluate nurses' burnout and the current level of institutional preparedness.
Author contributions
All authors contributed equally and validated the final version of record.
Declarations
Conflicts Of Interests
The Authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Registration
No registration applicable.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethical approval
Ethical approval for this study was not required.
References
1. Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus–Infected Pneumonia. N Engl J Med. 2020 Mar 26;382(13):1199–207. https://doi.org/10.1056/NEJMoa2001316
2. Al-Mandhari A, Samhouri D, Abubakar A, Brennan R. Coronavirus Disease 2019 outbreak: preparedness and readiness of countries in the Eastern Mediterranean Region. East Mediterr Health J. 2020 Feb 24;26(2):136–7. https://doi.org/10.26719/emhj.23.051
3. Novel Coronavirus (2019-nCoV): Situation Report 1 [Internet]. World Health Organization; 2020 Jan. Available from: https://apps.who.int/iris/bitstream/handle/10665/330776/nCoVsitrep31Jan2020-eng.pdf
4. Médecins Sans Frontières (MSF) [Internet]. 2021. Overlapping crises in Lebanon increase needs and worsen access to care. Available from: https://www.msf.org/lebanon-overlapping-crises-increase-needs-and-worsen-access-care
5. World Health Organization [Internet]. 2021. Weekly epidemiological update on COVID-19 – 23 March 2021. Available from: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---23-march-2021
6. Hackethal V. MedPage Today. 2021. India’s COVID-19 Variant: What We Know So Far. Available from: https://www.medpagetoday.com/special-reports/exclusives/92345
7. Guest JL, Del Rio C, Sanchez T. The three steps needed to end the COVID-19 pandemic: bold public health leadership, rapid innovations, and courageous political will. JMIR Public Health Surveill. 2020 Apr 6;6(2):e19043. https://doi.org/10.2196/19043
8. World Health Organization. Hospital Readiness Checklist for COVID-19 [Internet]. Copenhagen: WHO Regional Office for Europe; 2020. Available from: https://iris.who.int/bitstream/handle/10665/333972/WHO-EURO-2020-1012-40758-54954-eng.pdf
9. World Health Organization. Introduction to COVID-19: Methods for Detection, Prevention, Response and Control [Internet]. OpenWHO; 2021. Available from: https://openwho.org/courses/introduction-to-ncov
10. Saadeh D, Sacre H, Hallit S, Farah R, Salameh P. Knowledge, attitudes, and practices toward the coronavirus disease 2019 (COVID-19) among nurses in Lebanon. Perspect Psychiatr Care. 2021;57(3):1212–1221. https://doi.org/10.1111/ppc.12676
11. Domiati S, Itani M, Itani G. Knowledge, Attitude, and Practice of the Lebanese Community Toward COVID-19. Front Med. 2020, 7:542. https://doi.org/10.3389/fmed.2020.00542
12. Republic of Lebanon Ministry of Public Health (MoPH) [Internet]. 2020. New Website Launched by the Ministry of Information Regarding Coronavirus Latest News in Lebanon. Available from: https://www.moph.gov.lb/en/
13. Zhong BL, Luo W, Li HM, Zhang QQ, Liu ZG, Li WT et al. Knowledge, attitudes, and practices towards COVID-19 among Chinese residents during the rapid rise period of the COVID-19 outbreak: a quick online cross-sectional survey. Int J Biol Sci. 2020;16(10):1745–1752. https://doi.org/10.7150/ijbs.45221
14. Al-Hanawi MK, Angawi K, Alshareef N, Qattan AMN, Helmy HZ, Abudawood Y et al. Knowledge, Attitude and Practice Toward COVID-19 Among the Public in the Kingdom of Saudi Arabia: A Cross-Sectional Study. Front Public Health. 2020;8:217. https://doi.org/10.3389/fpubh.2020.00217
15. Haque T, Hossain K, Bhuiyan M, Ananna S, Mian M, Islam M, et al. Knowledge, attitude and practices (KAP) towards COVID-19 and assessment of risks of infection by SARS-CoV-2 among the Bangladeshi population: An online cross sectional survey [Internet]. medRxiv; 2020. Available from: https://www.medrxiv.org/content/10.1101/2020.05.26.20104497v1.full.pdf
16. Maheshwari S, Gupta PK, Sinha R, Rawat P. Knowledge, attitude, and practice towards coronavirus disease 2019 (COVID-19) among medical students: a cross-sectional study. J Acute Dis. 2020;9(3):100–104. https://doi.org/10.4103/2221-6189.283886
17. Zeenny RM, Ramia E, Akiki Y, Hallit S, Salameh P. Assessing knowledge, attitude, practice, and preparedness of hospital pharmacists in Lebanon towards COVID-19 pandemic: a cross-sectional study. J of Pharm Policy and Pract. 2020;13:54. https://doi.org/10.1186/s40545-020-00266-8
18. Wen X, Wang F, Li X, Gu H. Study on the Knowledge, Attitude, and Practice (KAP) of Nursing Staff and Influencing Factors on COVID-19. Front Public Health. 2021, 8:560606. https://doi.org/10.3389/fpubh.2020.560952
19. Lau LL, Hung N, Go DJ, Ferma J, Choi M, Dodd W et al. Knowledge, attitudes and practices of COVID-19 among income-poor households in the Philippines: a cross-sectional study. J Glob Health. 2020;10(1):011007. https://doi.org/10.7189/jogh.10.011007
20. Chaiban C, Maamari O, Issa ST, Kosremelli Asmar M: The experience of ER nurses in Lebanese hospitals during the COVID-19 outbreak: a qualitative study. Disaster Med Public Health Prep. 2023;17:e334. https://doi.org/10.1017/dmp.2022.304
21. Clinton M, Bou-Karroum K, Doumit MA, Richa N, Alameddine M. Determining levels of nurse burnout during the COVID-19 pandemic and Lebanon's political and financial collapse. BMC Nurs. 2022;21(1):11. https://doi.org/10.1186/s12912-021-00789-8
22. Youssef D, Abboud E, Abou-Abbas L, Hassan H, Youssef J. Prevalence and correlates of burnout among Lebanese health care workers during the COVID-19 pandemic: a national cross-sectional survey. J of Pharm Policy and Prac. 2022;15:102. https://doi.org/10.1186/s40545-022-00503-2
23. Bizri M, Kassir G, Tamim H, Kobeissy F, El Hayek S. Psychological distress experienced by physicians and nurses at a tertiary care center in Lebanon during the COVID-19 outbreak. J Health Psychol. 2022 May 1;27(6):1288–1300. https://doi.org/10.1177/1359105321991630